In this article, we will explore the intriguing connection between genetics and dementia. As our understanding of the causes of this debilitating condition deepens, researchers have found that genetics play a significant role in its development. By unraveling the genetic factors involved, scientists hope to shed light on potential treatments and preventative measures. Join us as we delve into the fascinating world of genetics and its impact on dementia.

Overview of Dementia
Dementia is a condition characterized by a decline in cognitive function that affects memory, thinking, behavior, and the ability to perform daily activities. It is not a specific disease but a syndrome caused by various underlying diseases or conditions. The most common form of dementia is Alzheimer’s disease, accounting for around 60-80% of all cases. Other types of dementia include vascular dementia, Lewy body dementia, frontotemporal dementia, and mixed dementia.
Definition of Dementia
Dementia is a broad term that encompasses a range of symptoms related to cognitive decline. It is characterized by the impairment of at least two cognitive domains, such as memory, language, attention, executive function, and visuospatial abilities. The decline in cognitive abilities is severe enough to interfere with daily activities and independence.
Types of Dementia
There are several different types of dementia, each with its own unique characteristics and causes. Alzheimer’s disease, as previously mentioned, is the most common type and is characterized by the accumulation of amyloid plaques and tau tangles in the brain. Vascular dementia is caused by reduced blood flow to the brain, often due to stroke or small vessel disease. Lewy body dementia is characterized by the presence of abnormal protein deposits called Lewy bodies in the brain. Frontotemporal dementia affects the frontal and temporal lobes of the brain, causing changes in personality and behavior. Mixed dementia refers to the presence of multiple types of dementia, commonly Alzheimer’s disease and vascular dementia.
Prevalence and Impact of Dementia
Dementia is a global issue that affects millions of people worldwide. According to the World Health Organization (WHO), there are approximately 50 million people living with dementia, and this number is expected to triple by 2050. The increasing prevalence of dementia has significant social, economic, and healthcare implications. Dementia places a significant burden on individuals, families, and society as a whole, often requiring long-term care and support. It is essential to understand the role of genetics in dementia to develop effective prevention and treatment strategies.
Genetic Factors in Dementia
While many factors contribute to the development of dementia, including age, lifestyle, and environmental factors, genetics also play a significant role. Numerous genetic variants and mutations have been identified that increase the risk of developing various types of dementia. Understanding these genetic factors is crucial for early detection, risk assessment, and personalized treatment plans.
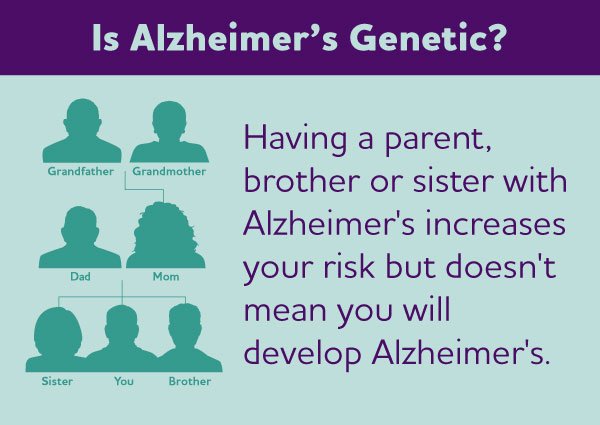
Alzheimer’s Disease Genetics
When it comes to genetics and dementia, Alzheimer’s disease genetics is one of the most well-studied areas. The APOE gene, specifically the APOE ε4 allele, is the most well-established genetic risk factor for late-onset Alzheimer’s disease. Individuals with one copy of the APOE ε4 allele have an increased risk of developing Alzheimer’s, while those with two copies have an even higher risk. Other genes, such as PSEN1, PSEN2, and APP, have been associated with early-onset familial Alzheimer’s disease but are considerably rarer.
Other Genetic Links to Dementia
In addition to Alzheimer’s disease, other genetic links to dementia have been identified. For example, mutations in the MAPT gene are associated with an increased risk of frontotemporal dementia. Mutations in the GRN gene have also been linked to frontotemporal dementia as well as corticobasal degeneration. Furthermore, mutations in the TREM2 gene have been associated with an increased risk of Alzheimer’s disease and other neurodegenerative disorders.
Autosomal Dominant Inheritance
Dementia can be inherited in different patterns, and one of the inheritance patterns seen in certain types of familial dementia is autosomal dominant inheritance. Autosomal dominant inheritance means that a single mutated copy of a gene from one affected parent is sufficient for an individual to inherit the condition. In the case of familial Alzheimer’s disease, which accounts for less than 5% of all Alzheimer’s cases, mutations in genes such as PSEN1, PSEN2, and APP can be inherited in an autosomal dominant manner.
Autosomal Recessive Inheritance
Autosomal recessive inheritance is another inheritance pattern seen in some cases of familial dementia. For a condition to be inherited in an autosomal recessive manner, both copies of the gene must be mutated, one inherited from each parent. In the context of dementia, autosomal recessive inheritance is less common but can occur in conditions such as Niemann-Pick disease type C, which can present with dementia symptoms.
Sporadic Inheritance
Not all cases of dementia have a clear genetic basis or follow strict inheritance patterns. Sporadic inheritance refers to cases where there is no known family history of the condition or where the genetic cause is not well understood. Sporadic forms of dementia, such as sporadic Alzheimer’s disease, are believed to be influenced by a combination of genetic, environmental, and lifestyle factors.

APOE Gene
The APOE gene, specifically the APOE ε4 allele, is one of the most well-known genetic risk factors for late-onset Alzheimer’s disease. The APOE ε4 allele is associated with an increased risk of developing Alzheimer’s and often leads to an earlier age of onset. However, it is important to note that not everyone with the APOE ε4 allele will develop Alzheimer’s, and not everyone without the allele will be free from the disease. The presence of the APOE ε4 allele is just one piece of the puzzle in understanding an individual’s risk for Alzheimer’s disease.
Other Susceptibility Genes
In addition to the APOE gene, other susceptibility genes have been identified that may contribute to the risk of developing dementia. For example, variations in genes such as CLU, CR1, and PICALM have been associated with an increased risk of late-onset Alzheimer’s disease. These genes are involved in various biological processes, including amyloid beta metabolism and inflammation, which are key mechanisms in the development of Alzheimer’s disease.
Mutations and Variants
Mutations and variants in various genes can contribute to the risk of developing dementia. These genetic changes can disrupt normal brain function, leading to the accumulation of toxic proteins, inflammation, and neuronal damage. While some mutations, such as those in genes associated with familial Alzheimer’s disease, have a clearly defined impact on disease risk, other variants may have a more subtle influence. Ongoing research aims to identify and characterize these genetic changes to better understand their impact on dementia development and progression.
Rare Genetic Variants in Familial Dementia
In some cases of familial dementia, rare genetic variants have been identified that significantly increase the risk of developing the condition. For example, mutations in the APP, PSEN1, and PSEN2 genes are associated with early-onset familial Alzheimer’s disease. These mutations disrupt the normal processing of amyloid beta protein, leading to the accumulation of amyloid plaques in the brain. Similarly, mutations in the MAPT and GRN genes are associated with frontotemporal dementia, altering the function of tau protein and leading to the formation of abnormal tau aggregates.
Rare Variants in Late-Onset Dementia
While rare variants are more commonly associated with early-onset familial forms of dementia, they can also play a role in late-onset dementia. Whole-genome sequencing studies have identified rare coding variants in genes such as SORL1, ABCA7, and TREM2 that increase the risk of late-onset Alzheimer’s disease. These variants may interact with other genetic and environmental factors to influence disease onset and progression.
Role of Rare Variants in Disease Progression
Rare genetic variants not only contribute to the risk of developing dementia but also play a role in disease progression. For example, certain genetic variants have been associated with a faster rate of cognitive decline or an increased likelihood of developing other complications associated with dementia. Understanding the impact of these rare variants on disease progression can help inform prognosis and personalized treatment strategies.
Diagnostic Genetic Testing
Diagnostic genetic testing involves analyzing an individual’s DNA to identify specific genetic changes associated with a known genetic condition. In the context of dementia, diagnostic genetic testing may be beneficial for individuals with a strong family history of dementia or symptoms suggestive of a specific genetic form of dementia, such as familial Alzheimer’s disease. Diagnostic genetic testing can help confirm a diagnosis, guide treatment decisions, and provide important information for family members who may also be at risk.
Predictive Genetic Testing
Predictive genetic testing is performed in individuals without symptoms to assess their risk of developing a genetic condition in the future. In the case of dementia, predictive genetic testing can provide information about an individual’s risk of developing certain types of dementia, such as Alzheimer’s disease. However, it is important to consider the psychological and emotional implications of such testing, as well as the limitations in predicting disease onset and progression based solely on genetic factors.
Ethical Considerations
Genetic testing for dementia raises several ethical considerations that need to be carefully addressed. These include issues of privacy, confidentiality, informed consent, and the potential impact of genetic information on individuals and their families. Genetic counselors and healthcare professionals play a crucial role in ensuring that individuals considering genetic testing for dementia have access to accurate information, support, and guidance.
Role of Genetic Counselors
Genetic counselors are healthcare professionals with specialized training in genetics and counseling. They play a vital role in providing accurate and comprehensive information about genetic conditions, including dementia, and helping individuals and families make informed decisions about genetic testing and their healthcare. Genetic counselors can assess an individual’s personal and family history, provide genetic education, discuss the implications of genetic testing, and offer psychosocial support.

Benefits of Genetic Counseling
Genetic counseling for dementia offers several benefits to individuals and families. It provides a safe and supportive environment to discuss concerns, understand the genetic basis of dementia, assess the risks and benefits of genetic testing, and explore available prevention and treatment options. Genetic counseling can also help individuals cope with the emotional and psychological aspects of living with or being at risk for dementia.
Support and Guidance
Living with or caring for someone with dementia can be challenging, both emotionally and physically. Genetic counseling can provide valuable support and guidance throughout the journey. Counselors can connect individuals and families with additional resources, support groups, and community organizations specializing in dementia care. They can also help navigate the complex healthcare system, coordinate care, and facilitate communication with other healthcare providers.
Genome-Wide Association Studies
Genome-wide association studies (GWAS) have revolutionized our understanding of the genetic basis of complex diseases, including dementia. These studies involve comparing the genomes of thousands of individuals with a specific condition, such as Alzheimer’s disease, to those without the condition. By identifying common genetic variants that are more prevalent in individuals with the condition, GWAS have helped identify new genes and pathways implicated in dementia.
Targeted Therapies
The identification of genes and genetic variants associated with dementia has opened up new avenues for targeted therapies. Targeted therapies aim to modify disease progression by targeting specific genetic mechanisms that contribute to the development and progression of dementia. For example, drugs targeting amyloid beta or tau proteins in Alzheimer’s disease are currently being investigated in clinical trials. Targeted therapies offer the potential for more personalized and effective treatments for individuals with specific genetic forms of dementia.
Precision Medicine Approaches
Precision medicine is an emerging field that aims to tailor medical treatments and interventions to individual characteristics, including genetic factors. In the context of dementia, precision medicine approaches leverage genetic information to identify the most effective treatments for a particular individual. By considering an individual’s genetic profile, healthcare providers can select the most appropriate medications, dosages, and interventions to maximize therapeutic benefits and minimize adverse effects.
Pharmacogenetics in Dementia Treatment
Pharmacogenetics is a branch of genetics that focuses on how genetic variations influence an individual’s response to medications. In the context of dementia treatment, pharmacogenetics can help identify individuals who are more likely to experience a positive response to specific medications or who may be at higher risk of adverse drug reactions.
Individualized Drug Response
Pharmacogenetic testing can help determine the most effective medications and dosages for individuals with dementia. By analyzing key genetic variations, healthcare providers can gain insights into an individual’s drug metabolism, drug-metabolizing enzyme activity, and drug target interactions. This information can guide treatment decisions and reduce the risk of adverse drug reactions.
Drug Side Effects
Dementia medications can have side effects that vary from person to person. Pharmacogenetic testing can identify individuals who may be at higher risk of experiencing certain side effects based on their genetic profile. This information can help healthcare providers select medications with a lower risk of adverse effects or adjust dosages to minimize side effects.
Optimizing Treatment
Pharmacogenetic testing offers the potential to optimize treatment outcomes for individuals with dementia. By personalizing medication selection and dosages based on an individual’s genetic profile, healthcare providers can improve treatment response, enhance quality of life, and potentially reduce healthcare costs associated with trial-and-error approaches.
Genetic Counseling and Prevention Strategies
In addition to genetic testing and personalized treatment approaches, genetic counseling can also play a crucial role in dementia prevention strategies.
Early Intervention and Risk Reduction
Early intervention is key in managing and potentially slowing the progression of dementia. Genetic counseling can help individuals and families identify early warning signs, understand their risk factors, and make lifestyle modifications to reduce their risk of developing dementia or delay its onset. This may include engaging in regular physical exercise, maintaining a healthy diet, managing chronic conditions such as hypertension and diabetes, and staying mentally and socially active.
Lifestyle Modifications
While genetics can influence an individual’s risk of developing dementia, lifestyle factors also play a significant role. Genetic counseling can provide guidance on lifestyle modifications that may help reduce the risk or delay the onset of dementia. This includes adopting a healthy diet, engaging in regular physical exercise, managing stress, getting enough sleep, and maintaining strong social connections.
Clinical Trials
Clinical trials are essential for advancing our understanding of dementia and developing new treatments. Genetic counseling can aid in identifying individuals who may be eligible to participate in clinical trials based on their genetic profile and specific genetic forms of dementia. Genetic counselors can explain the potential risks and benefits of participating in clinical trials and help individuals make informed decisions about their involvement.
In conclusion, understanding the role of genetics in dementia is crucial for early detection, risk assessment, and personalized treatment strategies. Genetic factors, including rare variants and mutations, contribute to the risk and progression of various types of dementia. Genetic testing and counseling provide valuable information and support to individuals and families affected by dementia, assisting in diagnosis, prevention, and treatment. As genetic research advances, it holds the promise of targeted therapies, precision medicine approaches, and improved outcomes for individuals living with dementia.
