Are you curious about the latest developments in Ewing Sarcoma research? Look no further! In this article, we will explore the most recent advancements in the understanding and treatment of this rare bone cancer. From groundbreaking discoveries to innovative therapies, you’ll discover how scientists and doctors are working tirelessly to improve the lives of those affected by Ewing Sarcoma. So, sit back, relax, and join us on this exciting journey through the cutting-edge world of Ewing Sarcoma research!

Introduction to Ewing Sarcoma
Background
Ewing Sarcoma is a rare type of cancer that primarily affects children and young adults. It was first described by Dr. James Ewing in 1921 and is characterized by the formation of tumors in the bones or soft tissues. The exact cause of Ewing Sarcoma is still unknown, but it is believed to be a result of genetic mutations or chromosomal rearrangements.
Epidemiology
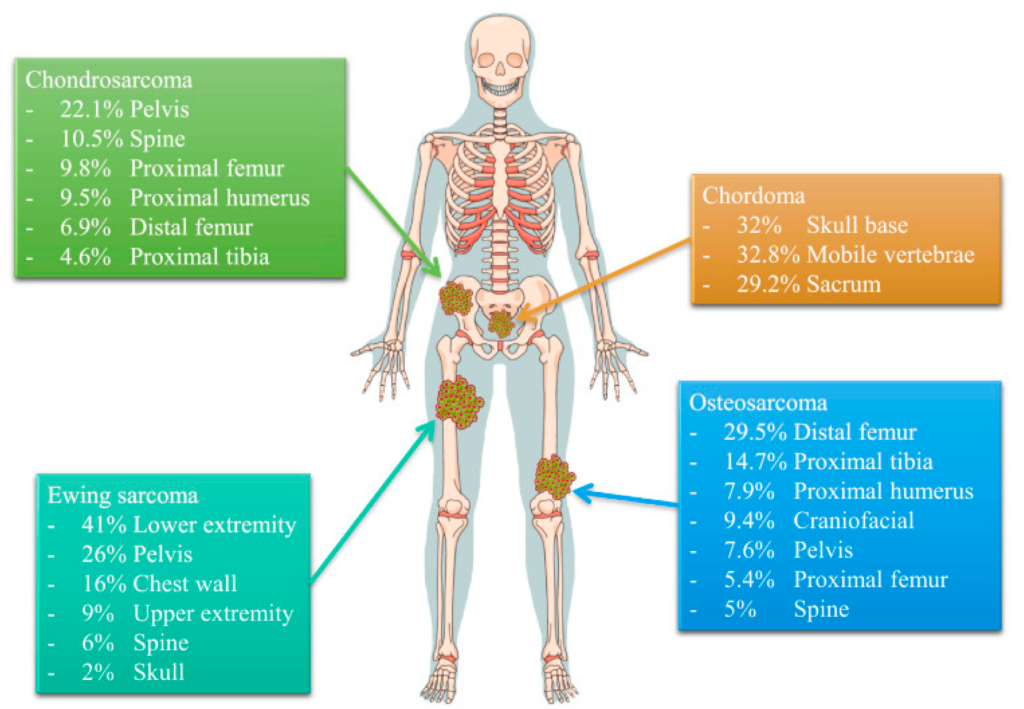
Ewing Sarcoma accounts for approximately 2% of all childhood cancers and typically occurs in the age range of 10 to 20 years. It is more common in males than females, with a ratio of about 1.6:1. While it can affect any race or ethnicity, it seems to be slightly more prevalent in Caucasians. The incidence of Ewing Sarcoma peaks during adolescence, and it is rare in adults over the age of 30.
Clinical Presentation
The clinical presentation of Ewing Sarcoma can vary depending on the location of the tumor. The most common symptom is pain, which may be localized or present as a deep ache. Swelling or a lump may also be noticed in the affected area. In some cases, the tumor may cause a fracture or lead to restricted movement. If Ewing Sarcoma has metastasized to other parts of the body, symptoms such as fatigue, weight loss, and bone pain in other areas may occur.
Diagnostic Methods
Imaging Techniques
Various imaging techniques are used to diagnose and stage Ewing Sarcoma. X-ray imaging is typically the first step and can reveal bone abnormalities such as tumors, fractures, or bone destruction. Magnetic Resonance Imaging (MRI) is often employed to assess the extent and location of the tumor and help with treatment planning. Computed Tomography (CT) scans and Positron Emission Tomography (PET) scans may also be used to evaluate the spread of the disease to other parts of the body, such as the lungs or lymph nodes.
Biopsy and Tumor Analysis
A biopsy is crucial for confirming the diagnosis of Ewing Sarcoma. A small sample of the tumor is obtained and examined under a microscope by a pathologist. The biopsy can be done through a needle (needle biopsy) or by surgically removing a portion of the tumor (incisional biopsy). In addition to identifying the presence of Ewing Sarcoma cells, further testing is usually performed to determine the genetic and molecular characteristics of the tumor, which can help guide treatment decisions.

Molecular Characterization of Ewing Sarcoma
Identification of EWSR1-ETS Fusion Proteins
Ewing Sarcoma is characterized by a specific genetic abnormality known as EWSR1-ETS fusion. This fusion occurs when a piece of the EWSR1 gene on chromosome 22 combines with a member of the ETS family of genes, typically FLI1 or ERG, on chromosome 11 or 21. The resulting fusion protein plays a critical role in the development of Ewing Sarcoma and serves as a unique marker for the disease.
Genomic Alterations
In addition to the EWSR1-ETS fusion, studies have identified other genomic alterations that contribute to the development and progression of Ewing Sarcoma. These alterations can involve changes in the number of copies of certain genes or the presence of specific mutations. Understanding these genomic alterations provides insights into the underlying biology of the disease and may lead to the identification of new therapeutic targets.
Epigenetic Modifications
Epigenetic modifications, such as DNA methylation and histone modifications, play a significant role in regulating gene expression and can influence the development of Ewing Sarcoma. Several studies have identified specific epigenetic changes associated with the disease, providing potential targets for therapy and aiding in the development of epigenetic-based treatment strategies.
Traditional Treatment Approaches
Chemotherapy
Chemotherapy is the cornerstone of treatment for Ewing Sarcoma. Combination chemotherapy regimens typically include drugs such as vincristine, doxorubicin, ifosfamide, and etoposide. These drugs work by targeting rapidly dividing cancer cells and can help shrink or eliminate tumors. Chemotherapy is usually administered in cycles over several months and can be given before or after surgery and radiation therapy.
Surgery
Surgery plays a crucial role in the management of Ewing Sarcoma. The goal of surgery is to remove the tumor and surrounding tissues while preserving as much function as possible. Depending on the location and extent of the tumor, surgical options may include a wide local excision, limb-sparing surgery, or amputation. In some cases, surgery may be necessary to remove metastatic tumors in other parts of the body.
Radiation Therapy
Radiation therapy uses high-energy beams to target and kill cancer cells. It is often used in combination with surgery and chemotherapy to improve local tumor control and reduce the risk of recurrence. The type and duration of radiation therapy depend on the location and size of the tumor. Radiation therapy can cause side effects, such as fatigue, skin changes, and potential long-term effects on growth and development, which should be carefully monitored and managed.

Resistance Mechanisms in Ewing Sarcoma
Multidrug Resistance Proteins
One major challenge in treating Ewing Sarcoma is the development of resistance to chemotherapy drugs. Multidrug resistance proteins, such as P-glycoprotein and multidrug resistance-associated protein 1 (MRP1), play a role in this resistance by effluxing drugs out of cancer cells, making them less effective. Understanding the mechanisms of drug resistance can help researchers develop new strategies to overcome this obstacle and improve treatment outcomes.
Altered DNA Repair Pathways
Ewing Sarcoma cells can develop alterations in DNA repair pathways, such as homologous recombination and non-homologous end joining, which can contribute to treatment resistance. These alterations enable cancer cells to repair DNA damage caused by chemotherapy or radiation, leading to decreased treatment efficacy. Targeting these altered DNA repair pathways may hold promise for enhancing the sensitivity of Ewing Sarcoma cells to therapy.
Immune-Based Therapies
Immunotherapy
Immunotherapy is a rapidly advancing field in cancer treatment, and Ewing Sarcoma is no exception. Immune-based therapies harness the power of the immune system to recognize and eliminate cancer cells. Approaches such as immune checkpoint inhibitors, chimeric antigen receptor (CAR) T-cell therapy, and immune-stimulating cytokines are being explored as potential treatments for Ewing Sarcoma. Early clinical trials have shown promising results, and further research is underway to evaluate the efficacy and safety of these treatments.
Targeting Checkpoint Inhibitors
Checkpoint inhibitors are a type of immunotherapy that involve blocking molecules called checkpoint proteins, such as PD-1 and CTLA-4, to unleash the immune system’s ability to recognize and destroy cancer cells. Preclinical studies and early-phase clinical trials have demonstrated the potential of checkpoint inhibitors in Ewing Sarcoma, particularly in combination with other treatment modalities. Ongoing research aims to optimize the use of these inhibitors to improve patient outcomes.
Adoptive Cell Transfer
Adoptive cell transfer involves the extraction, modification, and infusion of a patient’s own immune cells to enhance their ability to fight cancer. CAR T-cell therapy, a type of adoptive cell transfer, has shown promise in the treatment of certain types of leukemia and lymphoma, and efforts are underway to develop CAR T-cell therapies specifically for Ewing Sarcoma. This personalized approach holds potential for targeted and effective treatment.
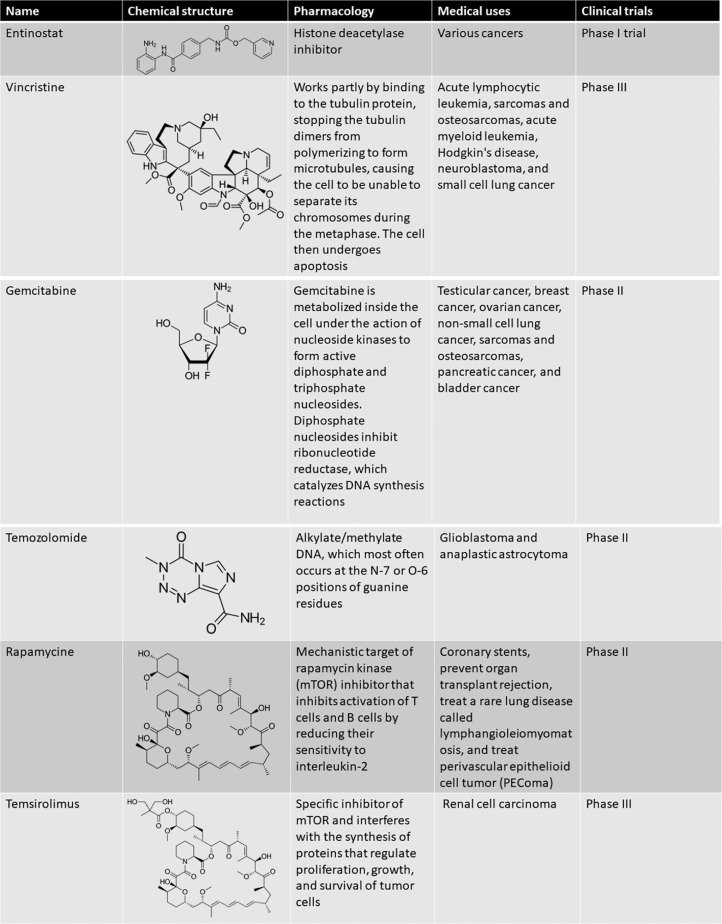
Emerging Targeted Therapies
Small Molecule Inhibitors
Small molecule inhibitors are drugs that target specific molecules involved in the development and growth of cancer cells. In Ewing Sarcoma, several small molecule inhibitors are under investigation, such as tyrosine kinase inhibitors (TKIs), mTOR inhibitors, and PARP inhibitors. These inhibitors can disrupt signaling pathways and cellular processes that are essential for cancer cell survival, potentially leading to tumor regression and improved patient outcomes.
Tyrosine Kinase Inhibitors
Tyrosine kinase inhibitors (TKIs) are a class of small molecule inhibitors that target enzymes called tyrosine kinases, which play a crucial role in cell signaling. Clinical trials are evaluating the efficacy of TKIs in Ewing Sarcoma, either as single agents or in combination with chemotherapy. Preliminary results suggest that TKIs may have a role in the treatment of Ewing Sarcoma, particularly in cases with specific genetic alterations.
Protein Degradation
Protein degradation is an emerging therapeutic approach that aims to selectively remove disease-causing proteins from cells. Proteolysis-targeting chimeras (PROTACs) and other protein degraders are being explored as potential treatments for Ewing Sarcoma. By targeting specific proteins that are critical for the survival or growth of cancer cells, protein degradation offers a novel strategy to disrupt tumor development and overcome treatment resistance.
Advances in Immunogenetics
Identification of Tumor Antigens
Tumor antigens are proteins expressed by cancer cells that can be recognized and targeted by the immune system. Advances in immunogenetics have led to the identification of specific tumor antigens associated with Ewing Sarcoma, including NY-ESO-1 and MAGE-A. These antigens have the potential to serve as targets for immunotherapies and personalized vaccines, as they are specifically expressed by cancer cells and not normal tissues.
Development of Personalized Vaccines
Personalized vaccines, also known as therapeutic cancer vaccines, are designed to stimulate the immune system to recognize and destroy cancer cells. In Ewing Sarcoma, personalized vaccines can be developed based on the individual patient’s tumor antigens and genetic profile. Clinical trials assessing the safety and efficacy of personalized vaccines are underway, and early results are encouraging. Personalized vaccines hold promise as a targeted and tailored approach to treating Ewing Sarcoma.
Precision Medicine in Ewing Sarcoma
Genomic Profiling
Genomic profiling involves analyzing the genetic makeup of a tumor to identify specific alterations that can be targeted with therapy. In Ewing Sarcoma, genomic profiling is increasingly being employed to identify actionable mutations or gene fusions that can guide treatment decisions. This personalized approach allows for the selection of targeted therapies that are more likely to be effective for each patient, improving treatment outcomes.
Targeted Therapy Selection
Targeted therapies aim to specifically inhibit molecules or pathways that are critical for tumor growth and survival. With advances in molecular characterization and genomic profiling, targeted therapies are being explored as potential options for Ewing Sarcoma. The identification of specific genetic alterations, such as EWSR1-FLI1 fusion, can guide the selection of targeted therapies that can interfere with the abnormal signaling pathways involved in Ewing Sarcoma development.
Long-Term Survival and Survivorship
Improved Outcomes
Over the years, advancements in diagnostic techniques, treatment approaches, and supportive care have led to improved outcomes for patients with Ewing Sarcoma. The combination of chemotherapy, surgery, and radiation therapy has been successful in achieving local control and long-term survival in many cases. Collaborative efforts among researchers and healthcare professionals continue to refine treatment strategies and enhance overall patient care.
Late Effects and Monitoring
While long-term survival rates have improved, survivors of Ewing Sarcoma may experience late effects related to their treatment. These can include cardiovascular issues, growth and development concerns, secondary cancers, and fertility problems. Regular monitoring and follow-up care are essential to identify and manage these late effects, ensuring that survivors receive appropriate medical attention and supportive services to optimize their quality of life.
In conclusion, Ewing Sarcoma is a challenging disease that requires a multidisciplinary approach for diagnosis, treatment, and survivorship care. Advances in molecular characterization, immunogenetics, and targeted therapies are offering new possibilities for personalized and more effective treatment strategies. With ongoing research and collaborative efforts, the future for patients with Ewing Sarcoma looks promising, bringing hope for improved outcomes and enhanced survivorship.