In this article, we will explore the recent breakthroughs in antiviral strategies that have been developed to combat influenza pandemics in humans. With the ever-present threat of influenza outbreaks, scientists and researchers have been working tirelessly to develop effective methods of preventing and treating this highly contagious virus. Through innovative approaches such as antiviral drugs, vaccines, and immune-boosting therapies, significant progress has been made in the fight against influenza. Join us as we delve into the exciting world of antiviral strategies and discover the latest advancements in influenza prevention and treatment.

Viral structure and characteristics
Overview of the influenza virus
The influenza virus, commonly known as the flu, is a highly contagious respiratory illness that affects millions of people worldwide each year. It belongs to the Orthomyxoviridae family and is classified into three main types: influenza A, B, and C. Influenza A is the most common and has the potential to cause pandemics due to its ability to undergo rapid genetic changes.
Structure of the influenza virus
The influenza virus is enveloped and spherical in shape, with a diameter of approximately 80-120 nanometers. It consists of an outer lipid membrane that surrounds the viral core. The viral core contains the viral RNA, which is segmented into eight separate RNA strands. The surface of the virus is covered with two types of glycoproteins: hemagglutinin (HA) and neuraminidase (NA). These proteins play crucial roles in viral attachment and entry into host cells.
Genetic variability of influenza viruses
One of the defining characteristics of the influenza virus is its ability to undergo frequent mutations and reassortments. This genetic variability is responsible for the emergence of new strains and the continuous need for updated vaccines. Antigenic drift, which involves gradual mutations in the surface proteins, leads to seasonal epidemics. Antigenic shift, on the other hand, occurs when two different influenza A viruses infect the same host and exchange genetic material, resulting in the emergence of a novel strain with pandemic potential.
Modes of influenza transmission
Airborne transmission
The influenza virus is primarily transmitted through respiratory droplets that are expelled when an infected person coughs, sneezes, or talks. These droplets can travel short distances and infect others who come into close contact with the infected individual. Airborne transmission can also occur when these droplets remain suspended in the air for extended periods, especially in enclosed spaces with poor ventilation.
Direct contact transmission
Direct contact with respiratory secretions, such as touching contaminated surfaces or shaking hands with an infected person, can also lead to influenza transmission. When infected individuals touch their nose or mouth and then touch surfaces or objects, they can leave behind the virus, which can survive for a short period outside the body. Healthy individuals who touch these contaminated surfaces and then touch their own eyes, nose, or mouth can become infected with the virus.
Indirect contact transmission
Influenza can also be transmitted indirectly through the contamination of objects or surfaces. The virus can survive on surfaces such as doorknobs, countertops, or toys, and remain infectious for several hours. When an individual touches these contaminated surfaces and then touches their face, the virus can enter the body through the eyes, nose, or mouth, leading to infection.
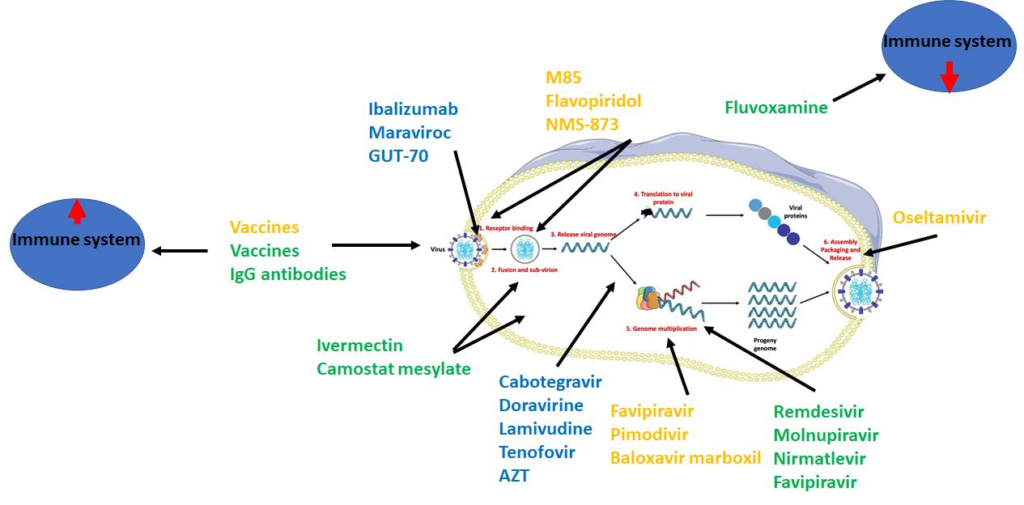
Antiviral drugs for influenza treatment
Neuraminidase inhibitors
Neuraminidase inhibitors are a class of antiviral drugs that target the neuraminidase protein on the surface of the influenza virus. These drugs, such as oseltamivir (Tamiflu) and zanamivir (Relenza), work by inhibiting the release of newly formed virus particles from infected cells. Neuraminidase inhibitors have been shown to reduce the severity and duration of influenza symptoms, but their efficacy is dependent on the timing of administration.
Adamantanes
Adamantanes, including amantadine and rimantadine, are another class of antiviral drugs used for the treatment of influenza A infections. These drugs inhibit the M2 ion channel protein, which is involved in the uncoating process of the virus inside host cells. However, widespread resistance to adamantanes has emerged in recent years, limiting their clinical usefulness.
M2 ion channel inhibitors
M2 ion channel inhibitors, such as amantadine and rimantadine, target the M2 protein of the influenza virus. These drugs prevent the release of viral genetic material into host cells, thereby inhibiting viral replication. However, similar to adamantanes, resistance to M2 ion channel inhibitors has become widespread, and their use is now limited.
Host immune response to influenza infection
Innate immune response
The innate immune response is the first line of defense against influenza infection. It involves the activation of various immune cells, such as macrophages and natural killer cells, which recognize and eliminate the virus. Additionally, the innate immune response triggers the release of pro-inflammatory molecules, such as cytokines, to recruit immune cells and enhance the adaptive immune response.
Adaptive immune response
The adaptive immune response is a more targeted and specific immune response that develops after the initial innate immune response. It involves the activation and proliferation of B cells and T cells, which produce antibodies and target infected cells, respectively. The adaptive immune response plays a crucial role in clearing the virus and providing long-lasting immunity.
Cross-reactive immunity
Cross-reactive immunity refers to the ability of the immune system to recognize and respond to different strains of influenza virus, even if the individual has not been previously exposed to that particular strain. This phenomenon occurs due to shared antigenic sites between different strains of the virus. Cross-reactive immunity can provide partial protection against new strains and contribute to milder disease outcomes.
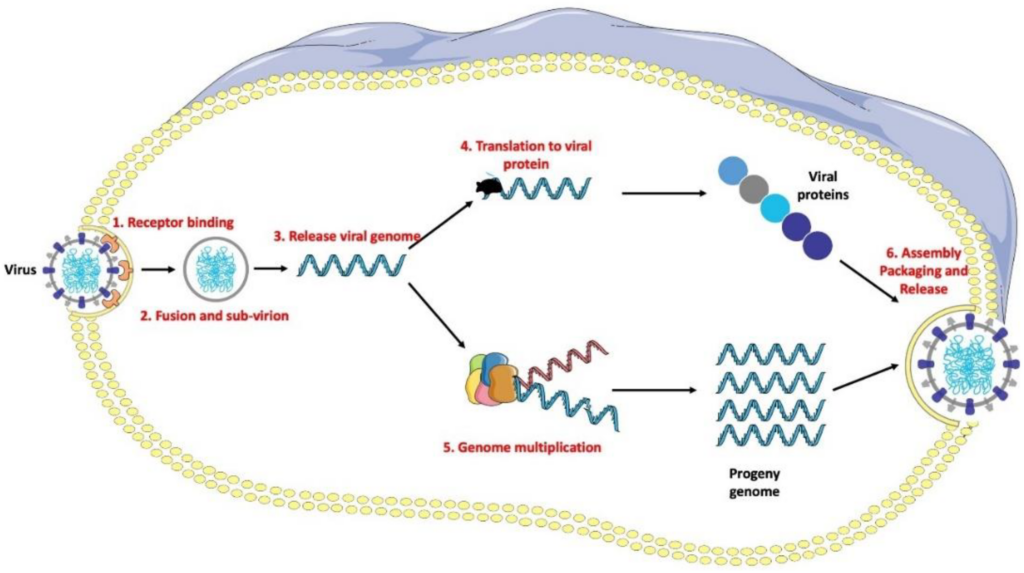
Vaccination strategies against influenza
Inactivated influenza vaccines
Inactivated influenza vaccines, also known as flu shots, contain killed or inactivated influenza virus components. These vaccines are typically administered via injection and stimulate the immune system to produce an immune response against the virus. Inactivated vaccines are effective against specific strains included in the vaccine, and their composition is updated yearly to match the circulating strains.
Live attenuated influenza vaccines
Live attenuated influenza vaccines contain weakened forms of the influenza virus that are unable to cause severe disease. These vaccines are usually administered as a nasal spray and stimulate both innate and adaptive immune responses. Live attenuated vaccines offer the advantage of closely mimicking natural infection, providing a broader immune response that may confer better protection against circulating strains.
Recombinant influenza vaccines
Recombinant influenza vaccines are produced by genetically engineering the virus or viral proteins. These vaccines do not require the use of eggs and can be manufactured more quickly compared to traditional methods. Recombinant vaccines mainly target the viral surface protein, hemagglutinin, and induce a specific immune response against the virus. They have shown promising results in terms of efficacy and manufacturing speed.
Challenges in antiviral treatment and prevention
Antiviral resistance
One of the major challenges in influenza treatment is the emergence of antiviral resistance. Over time, the influenza virus can acquire mutations that make it less susceptible to the effects of antiviral drugs. This resistance can limit the effectiveness of available treatment options and necessitate the development of new antiviral drugs. Regular surveillance of circulating strains is essential to monitor antiviral resistance patterns.
Vaccine strain selection
Each year, the World Health Organization (WHO) collaborates with experts in virology and epidemiology to determine the strains that are most likely to circulate during the upcoming flu season. This strain selection process is based on global surveillance data and aims to provide the best possible match between the vaccine and circulating strains. However, occasionally, the selected strains may not perfectly match the circulating strains, reducing the effectiveness of the vaccine.
Annual vaccine production
The production of influenza vaccines is a complex and time-consuming process. It involves growing the virus in eggs or cell cultures, purifying the viral components, and formulating the vaccine. This process can take several months, limiting the flexibility to rapidly respond to emerging strains or unexpected outbreaks. Advances in vaccine production technologies, such as cell-based or recombinant methods, have the potential to shorten the production timeline and improve vaccine availability.
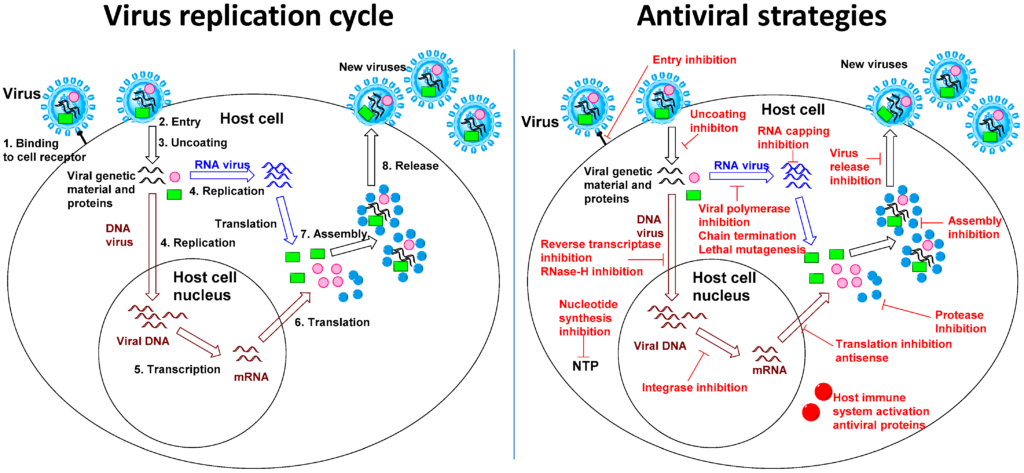
Emerging antiviral strategies
Broad-spectrum antivirals
Broad-spectrum antivirals are a new class of antiviral agents that target conserved regions of the viral genome or viral proteins. These drugs have the potential to be effective against multiple strains and types of the influenza virus. Examples of broad-spectrum antivirals under development include fusion inhibitors, polymerase inhibitors, and viral entry inhibitors. The development of broad-spectrum antivirals could address the challenges associated with antiviral resistance.
Nanotechnology-based approaches
Nanotechnology offers innovative solutions for the prevention and treatment of influenza. Nanoparticles can be engineered to deliver antiviral drugs directly to the site of infection, enhancing their efficacy while minimizing side effects. Furthermore, nanomaterials can be used to develop rapid diagnostic tests for influenza, enabling early detection and prompt treatment. Nanotechnology-based approaches hold promise for improving both prevention and treatment strategies against influenza.
Gene therapy for influenza
Gene therapy is a cutting-edge approach that aims to manipulate the expression of specific genes to prevent or treat diseases. In the context of influenza, gene therapy could involve modifying the expression of immune-related genes to enhance the host’s ability to combat the virus. Additionally, gene therapy could potentially be used to directly target and inhibit viral replication. Although gene therapy for influenza is still in the early stages of development, it represents a promising future direction for antiviral strategies.
Targeting host factors in the infectious process
Host proteases as therapeutic targets
Host proteases play a crucial role in the replication and spread of the influenza virus within the host. By targeting these proteases with specific inhibitors, it may be possible to disrupt the viral life cycle and prevent infection. Several protease inhibitors, such as camostat mesylate, have shown potential in preclinical studies. Targeting host proteases provides an alternative approach to antiviral treatment by focusing on host factors rather than directly targeting the virus.
Modulation of host immune response
In addition to targeting the virus, modulating the immune response of the host can also be an effective strategy against influenza. By manipulating immune signaling pathways, it may be possible to enhance the early innate immune response and promote rapid viral clearance. Various immune-modulating agents, such as interferons and immune checkpoint inhibitors, are currently being studied for their potential in limiting influenza severity and improving patient outcomes.
Targeting host lipid metabolism
Host lipid metabolism is a critical factor in the replication and assembly of the influenza virus. Disrupting lipid metabolism pathways in the host could potentially inhibit viral replication and spread. Several lipid-lowering drugs, such as statins, have been shown to have antiviral effects against influenza in preclinical studies. Targeting host lipid metabolism represents an intriguing approach to both treatment and prevention strategies against influenza.

Innovative vaccine design and delivery approaches
Universal influenza vaccines
Universal influenza vaccines aim to provide broad and long-lasting protection against a wide range of influenza strains, including those that have not yet emerged. These vaccines target conserved regions of the virus that are less prone to mutation. Strategies such as nanoparticle-based vaccines and viral vector-based vaccines are being explored to enhance the immunogenicity and breadth of protection of universal influenza vaccines.
Next-generation vaccine platforms
Next-generation vaccine platforms utilize advanced techniques to improve vaccine efficacy and production. For example, DNA-based vaccines involve the direct delivery of viral DNA into host cells, triggering an immune response. Other platforms include virus-like particles (VLPs) that mimic the structure of the virus without the genetic material. These next-generation vaccine platforms have the potential to enhance vaccine immunogenicity, simplify production processes, and enable rapid response to emerging strains.
Alternative routes of vaccine administration
Traditional influenza vaccines are administered through injection, but alternative routes of administration are also being explored. Intranasal vaccination, for example, involves delivering the vaccine directly into the respiratory system, which is the site of infection. This approach may enhance mucosal immunity and provide better protection against respiratory pathogens. Other alternative routes, such as transdermal patches or oral vaccines, offer convenience and may improve vaccine uptake.
Conclusion and future perspectives
Advancements in antiviral strategies for influenza continue to evolve, addressing challenges in treatment and prevention. The development of broad-spectrum antivirals, nanotechnology-based approaches, gene therapy, and targeting host factors are promising avenues for future research. Furthermore, innovative vaccine design and delivery approaches, such as universal vaccines, next-generation platforms, and alternative administration routes, hold potential for improving vaccine effectiveness and accessibility. While there are still challenges to overcome, these advancements provide hope for better prevention and treatment of influenza in the future. With ongoing research and collaboration, breakthroughs in influenza prevention and treatment are within reach.